Why Easy Density Experiments Are Perfect for At-Home Science
Easy density experiments are one of the best ways to make science click for kids at home — no fancy lab required.
Here are some quick, crowd-pleasing options you can try today:
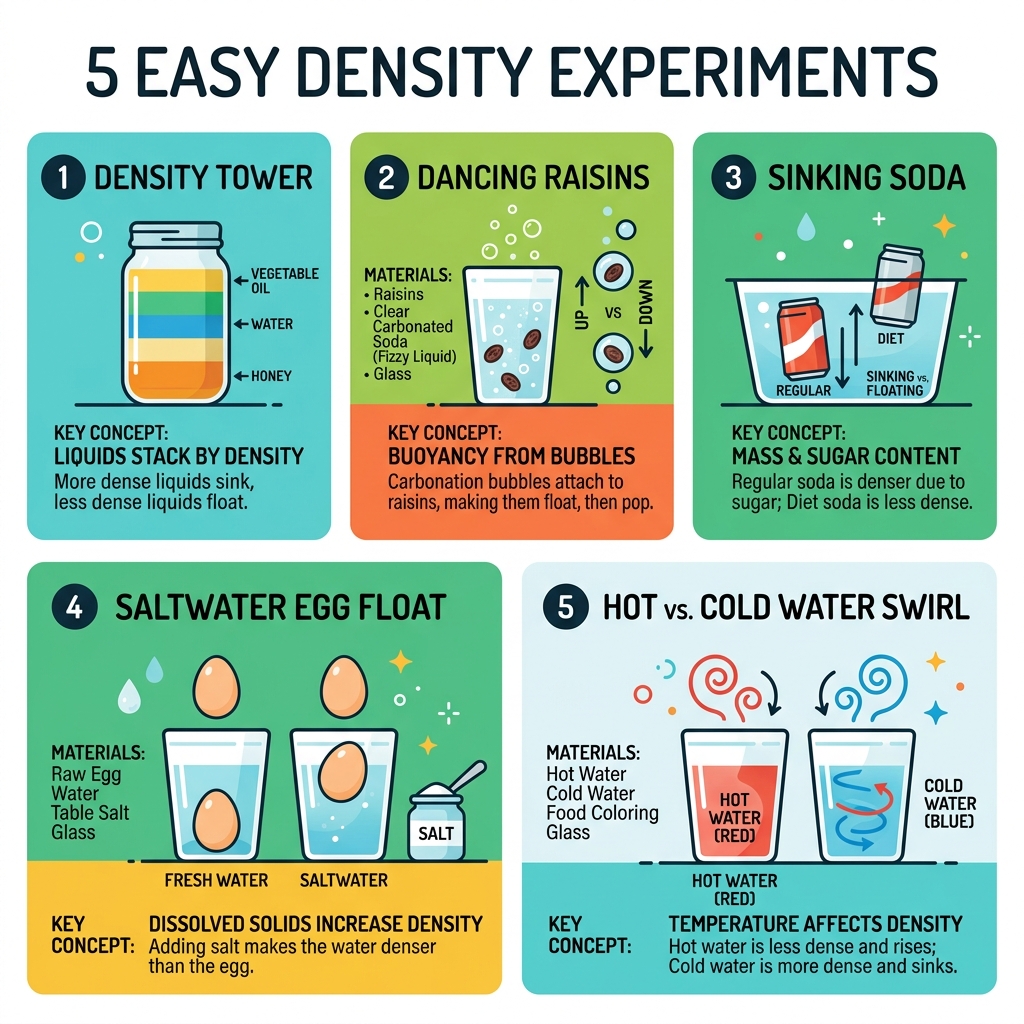
- Density Tower — Layer honey, corn syrup, dish soap, water, vegetable oil, and rubbing alcohol in a clear jar to create a stunning rainbow of liquids.
- Dancing Raisins — Drop raisins into a glass of Sprite and watch them rise and fall as carbonation bubbles attach and pop.
- Sinking Soda — Place a can of regular soda and a can of diet soda in a tub of water. One sinks, one floats — and kids love figuring out why.
- Saltwater Egg Float — Dissolve salt into water and watch a raw egg go from sinking to floating.
- Hot vs. Cold Water Swirl — Add food coloring to hot and cold water, then combine them slowly to see how temperature affects how liquids mix.
Density is one of those science topics that feels like magic the first time you see it. Why does oil always float on water? Why does a massive steel ship stay afloat while a small pebble sinks? It all comes down to density — how much mass is packed into a given space.
The best part? You don’t need a classroom or a science kit. Most of these experiments use things already sitting in your kitchen cupboard.
Whether you have five minutes or an afternoon, these activities are low-stress, high-impact, and genuinely fun — for kids and the grown-ups watching alongside them.
What is Density and Why Does It Matter?
At its simplest, density is a measure of how tightly “stuff” is packed into a space. Think of it like a drawer full of socks. If you have five pairs of socks loosely tossed in, the drawer isn’t very dense. But if you jam thirty pairs in there and have to sit on the drawer to close it, that drawer is now very dense! In science terms, we are looking at mass (the amount of matter or “stuff”) relative to volume (the amount of space it takes up).
Everything around us is made of atoms. In some objects, like a lead weight, the atoms are packed together like commuters on a crowded morning train. In others, like a marshmallow or a balloon, the atoms have plenty of room to wiggle around. This arrangement determines whether something will sink or float when placed in a liquid.
Understanding this concept isn’t just for passing a quiz; it helps us understand how the world works. For instance, Density Column Science explains why certain liquids, like oil and vinegar in salad dressing, refuse to stay mixed. In the real world, this principle is vital for environmental science. When an oil spill occurs in the ocean, the oil floats on top of the salt water because it is less dense. This allows scientists to develop skimmers to clean the surface, though it also explains why surface-dwelling wildlife is so heavily impacted.
Density also explains the massive movements of our planet. The Earth is layered like a giant version of our density jar: a super-dense solid iron core at the center, followed by a slightly less dense mantle, and finally the thin, least-dense crust we live on. Even the giant conveyor belts of ocean currents are driven by density; cold, salty water is denser and sinks, pushing warmer water across the globe. For parents looking to introduce these big ideas early, starting with first-steps-in-science-simple-experiments-for-kids is a great way to build that foundational curiosity.
The Science Behind Easy Density Experiments
When we drop an object into a liquid, two main forces are at play: gravity pulling it down and buoyancy pushing it up. This upward force is described by Archimedes’ principle, which states that any object, wholly or partially immersed in a fluid, is buoyed up by a force equal to the weight of the fluid displaced by the object.
If the object is denser than the liquid, it weighs more than the water it moves out of the way, so it sinks. If it is less dense, the water pushes it up with more force than gravity pulls it down, so it floats. Sometimes, an object is exactly as dense as the liquid, leading to neutral buoyancy — it just hovers in the middle! This is exactly how scuba divers use weights and air vests to stay suspended at a specific depth without sinking to the bottom or bobbing to the top.
Exploring these forces at home allows kids to visualize invisible physics. We often recommend starting with simple observation-based demos before diving into the math. You can find more inspiration for these “phenomena-first” activities at sparks-curiosity-science-experiments-for-kids-at-home.
How We Measure Density in Easy Density Experiments
In the lab (or your kitchen), density is usually measured in grams per milliliter (g/mL) for liquids or grams per cubic centimeter (g/cm³) for solids. The formula is simple: Density = Mass / Volume.
To find the density of a liquid at home, you can use a kitchen scale to find the mass and a measuring cup or a graduated cylinder to find the volume. A fun fact to remember is that the density of pure water is almost exactly 1 g/mL. This makes water the perfect “control” for our easy density experiments. Anything with a density higher than 1 will sink in water; anything lower than 1 will float.
If you’re ready to turn your kitchen into a calculation lab, we have a guide on engaging-minds-hands-on-stem-activities-at-home-for-kids that walks you through how to use basic household tools to get accurate measurements for your science projects.
Top 5 Easy Density Experiments to Try at Home
If you want to see a “wow” moment, the multi-layered density tower is the undisputed king of easy density experiments. It looks like a liquid rainbow trapped in a jar, and it perfectly demonstrates how different substances can occupy the same space without mixing.

To create a professional-looking 6-layer column, you will need:
- Honey (The densest)
- Corn syrup or Maple syrup
- Dish soap (usually blue or green)
- Water (dyed with food coloring)
- Vegetable oil
- Rubbing alcohol (dyed a different color than the water)
The secret to success is the layering order and the turkey baster technique. You must start with the densest liquid (honey) and work your way up. For a detailed step-by-step on the specific measurements, check out this guide on the STEM Live for Kids Experiment 3: 6-Layer Density Column.
When adding the lighter layers (like water, oil, and alcohol), don’t just dump them in! Use a turkey baster or a spoon to slowly dribble the liquid down the inside wall of the glass. This prevents the force of the falling liquid from breaking the surface tension of the layer below it. If you’re careful, you’ll have sharp, distinct lines between every color.
Sinking Soda and Dancing Raisins
One of our favorite “bet you didn’t know” experiments involves two cans of soda. Grab a can of regular Coke and a can of Diet Coke. Ask your kids: “Which one will float?” Most will guess both will sink because they are heavy metal cans. However, when you drop them in a tub of water, the regular soda sinks like a stone, while the diet soda bobs on the surface.
The reason? Sugar content. A regular can of soda has about 39 grams of sugar dissolved in it, which significantly increases its mass without changing its volume. Diet soda uses a tiny amount of artificial sweetener, which adds almost no mass. This makes the regular soda denser than water and the diet soda slightly less dense.
For a more active experiment, try Dancing Raisins (or “Dancing Pasta”). Fill a glass with a clear carbonated soda like Sprite. Drop in a few raisins or pieces of small, dry pasta. Initially, they sink because they are denser than the soda. But soon, bubbles of carbon dioxide gas (which is very low density) attach to the rough surface of the raisins. These bubbles act like tiny life jackets, increasing the volume of the raisin without adding much mass, which lowers the overall density and carries them to the top. When the bubbles pop at the surface, the density increases again, and they sink. It’s a rhythmic, bubbly ballet! We explore more about gas-based experiments at sparks-curiosity-science-experiments-for-kids-at-home-2.
Saltwater Magic and Temperature Swirls
You can actually change the density of water right before your eyes by adding dissolved substances or changing the temperature. In the Saltwater Egg Float, a raw egg will sink in a glass of plain tap water. But as you stir in spoonfuls of salt, you are packing more “stuff” (salt molecules) into the same volume of water. Eventually, the saltwater becomes denser than the egg, and the egg miraculously rises to the surface. This is why it’s so much easier for humans to float in the ocean than in a swimming pool!
Temperature also plays a huge role. In our Temperature Swirl experiment, we use hot and cold water dyed with different colors. Hot water molecules are agitated and move around quickly, pushing each other apart. This means there are fewer molecules in a cup of hot water than in a cup of cold water — making hot water less dense. If you carefully layer hot red water over cold blue water, they will stay separated for a while.
Interestingly, water behaves very strangely when it freezes. Most substances get denser when they turn solid, but water expands. It forms a hexagonal, honeycomb-like molecular structure that leaves a lot of empty space. This is why ice floats! If ice didn’t float, lakes would freeze from the bottom up, killing all the fish inside. You can find more “cool” facts about water behavior in the Quick & Easy Density resource or our guide on first-steps-in-science-simple-experiments-for-kids-2.
Advanced Fun: The Brazil Nut Effect and Lava Lamps
Once you’ve mastered liquids, it’s time to look at granular convection, also known as the “Brazil Nut Effect.” Have you ever noticed that in a container of mixed nuts, the large Brazil nuts always end up at the top? This is a form of density and size segregation. When you shake the container, the smaller nuts (like peanuts) fall into the tiny gaps created during the movement, eventually pushing the larger, less “efficiently packed” nuts to the surface. It’s a great way to show that density concepts apply to solids too!
Another advanced favorite is the Homemade Lava Lamp. This combines density with polarity. Oil and water are immiscible, meaning they won’t mix because water molecules are polar (like little magnets) and oil molecules are non-polar. Since oil is less dense, it sits on top. When you drop an Alka-Seltzer tablet into the mix, it reacts with the water at the bottom to create carbon dioxide bubbles. These bubbles stick to the water droplets and carry them up through the oil. At the top, the gas escapes, the water’s density “increases” (relative to the gas-water combo), and the droplet sinks back down.
For a parent’s perspective on managing these slightly messier (but worth it!) projects, take a look at no-lab-coat-required-simple-science-for-your-5-year-old.
Safety Tips for Home Science Labs
While easy density experiments are generally safe, we always recommend a few ground rules to keep the kitchen from turning into a disaster zone:
- Adult Supervision: Especially when using rubbing alcohol, food coloring (which stains!), or glass containers.
- Mark Your Containers: Some of these experiments look like delicious juice. Use a Sharpie to mark “SCIENCE – DO NOT DRINK” on any jars containing non-food items like soap or alcohol.
- Chemical Safety: Never mix cleaning solutions unless the experiment specifically calls for it. For example, mixing bleach and ammonia creates toxic gas. Stick to the household ingredients listed in the guides.
- Clean-Up: Honey and syrup are sticky! Have a damp cloth ready, and try to conduct experiments on a tray to catch spills.
Common Liquid Densities for Comparison
| Liquid | Approximate Density (g/mL) |
|---|---|
| Honey | 1.42 |
| Corn Syrup | 1.33 |
| Dish Soap | 1.06 |
| Water | 1.00 |
| Vegetable Oil | 0.92 |
| Rubbing Alcohol | 0.79 |
For more tips on organizing your home “lab” and making clean-up a breeze, check out engaging-minds-hands-on-stem-activities-at-home-for-kids-2.
Frequently Asked Questions about Density
Why does ice float in water but sink in rubbing alcohol?
Ice is a bit of a rebel. When water freezes, it expands, meaning the same mass takes up more space. This makes ice (0.92 g/mL) less dense than liquid water (1.00 g/mL), so it floats. However, rubbing alcohol is very “loose” and has a density of only about 0.79 g/mL. Since 0.92 is higher than 0.79, the ice is “heavier” than the alcohol it displaces, causing it to sink to the bottom.
What is the density of water?
The density of water is essentially 1 g/mL. This is not a coincidence; the metric system was actually designed around water! While temperature and salt can change this slightly, 1 g/mL is the standard we use for almost all elementary science.
Why does regular soda sink while diet soda floats?
It all comes down to the “stuff” inside. A 12-ounce can of regular soda contains roughly 10 teaspoons of sugar. Sugar is dense and adds significant mass to the liquid. Diet soda uses artificial sweeteners like aspartame, which are hundreds of times sweeter than sugar, so only a tiny, weightless amount is needed. This makes the total mass of the diet soda can less than the mass of the water it displaces.
Conclusion
At Curta Arte, we know that being a single parent means your time is precious. You want to provide your kids with the best educational experiences, but you don’t always have the bandwidth for a three-hour curriculum. That’s why we love easy density experiments. They are fast, they use what you have, and they spark those “lightbulb moments” that foster a lifelong love of learning.
As Aria James often says, science isn’t about having all the answers; it’s about asking the right questions. Whether you’re watching a raisin dance or building a six-story liquid tower, you’re doing more than just a “trick” — you’re building a bond and a brain.
For more ideas on how to keep the kids engaged without losing your mind, explore our other Fun activities for kids and join our community of parents making magic out of the everyday.